Key Releases are ad hoc announcements pursuant to SIX Swiss Exchange Article 53 Listing Rules.
Header
News Archive
News Archive Navigation
icon
News Archive Navigation Language
Language Preferences
Showing 1809 results
August 2018
-
Key Release
Novartis appoints Dr. Klaus Moosmayer as Chief Ethics Risk and Compliance Officer
Basel, August 14, 2018 - Novartis announced today the appointment of Dr Klaus Moosmayer as Chief Ethics, Risk and Compliance Officer. He will report to Vas Narasimhan, M.D., CEO of Novartis and… -
Story Discovery

New drug manufacturing tools change pharma chemistry
The modernization and miniaturization of pharmaceutical production could expand the range of ingredients medicinal chemists use to make new drugs.
-
Story Discovery

Embracing big data to understand complex diseases
Researchers discover that networks of proteins circulating in the blood influence the development and progression of chronic diseases.
-
Story Discovery

Discovery gives cystic fibrosis researchers new direction
Novartis-Harvard team identifies a new type of cell that is key to the disease.
July 2018
-
Story Access to Healthcare
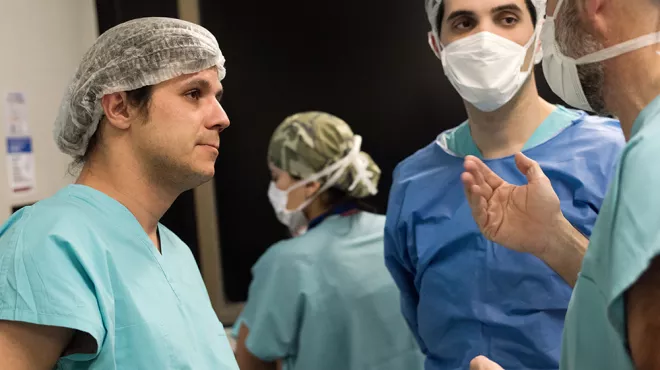
Meet Manuel Cobos, transplant surgeon and researcher
Follow Dr. Cobos as he works to provide a patient with a new liver and performs surgery using a tiny video camera.
-
Media Release
Novartis marks a new era for migraine patients with the EU approval of Aimovig®, a first-of-its-kind treatment specifically designed for migraine prevention
Patients on Aimovig (erenumab) in clinical trials reported consistent and sustained migraine prevention, with many experiencing a 50% or more reduction in monthly migraine days; safety and… -
Story Discovery

Quest for a new measure of mental health
Novartis researchers are searching for more effective ways to discover drugs for neurological and psychiatric diseases.
-
Media Release
Sandoz receives European Commission approval for biosimilar Hyrimoz® (adalimumab)
Biosimilar Hyrimoz® (adalimumab) approved for use in all same indications as reference medicine*** including rheumatology, gastroenterology and dermatology Early therapeutic intervention is… -
Media Release
Novartis combination Tafinlar® + Mekinist® receives positive CHMP opinion for adjuvant treatment of BRAF V600 mutation-positive melanoma
Phase III trial showed a 53% reduction in risk of recurrence or death with the combination of a BRAF and MEK inhibitor as adjuvant therapy versus placebo[1] Relapse-free survival benefit with… -
Media Release
Novartis renews drug donation of Egaten® (triclabendazole) until 2022, reaffirming its commitment to the fight against liver fluke
Novartis extends agreement with the WHO for the donation of Egaten® for the treatment of liver fluke, a neglected tropical disease, also known as fascioliasis, that infects more than 2.4 million… -
Media Release
Novartis demonstrates commitment to lead immuno-dermatology by in-licensing IL-17C compound for atopic dermatitis
Novartis enters an exclusive license agreement with Galapagos and MorphoSys for an investigational biologic compound, MOR106, a novel antibody directed against IL-17C IL-17C is believed to… -
Media Release
Novartis Kisqali® now first and only CDK4/6 inhibitor indicated in US as first-line therapy specifically for premenopausal women; and as initial therapy with fulvestrant in postmenopausal women
Kisqali is now the only CDK4/6 inhibitor indicated in combination with an aromatase inhibitor as first-line treatment for pre-, peri- or postmenopausal women with HR+/HER2- advanced breast cancer…
Pagination
- ‹ Previous page
- 1
- …
- 84
- 85
- 86
- 87
- 88
- 89
- 90
- …
- 151
- › Next page